Concept Question
The result of beta(-) decay of cesium-137 would be
(mouse over choices to get answer)
Types of Ionizing Radiation
The radiation emitted from decaying nuclei is called ionizing radiation. This is because it is sufficiently high in energy to ionize essentially any element or compound that it interacts with. This is the essential danger of radioactive materials. They are constantly undergoing decay and emitting ionizing radiation. That radiation will interact with molecules in your body and kick off electrons. This will in turn lead to undesired chemistry in your body (bonds breaking, the formation of free radicals, ...). The effects of exposure to the radiation can acute and similar to the effects of UV-radiation (like a sunburn only much much worse). Or they could be long term such as an increased risk of the development of certain cancers.
There are three types of ionizing radiation that were classified with generic names before they were completely characterized. These initial terms of alpha, beta, and gamma radiation are still used despite the fact that we now know specifically what all three are.
Alpha Radiation
Alpha radiation is a form of ionizing radiation that is associated with radioactive isotopes that undergo alpha decay. These isotopes decay with the emission of an alpha particle (He-4 nucleus).
Alpha particles have a very strong ionizing power (they are "missing" two electrons), but generally alpha radiation is easy to protect against since alpha particles have very little penetrating power due to their large mass. As such, they cannot pass through paper or even the dead layers of skin on a human body. However, alpha emitters can be very dangerous if they make their way into the body. In particular, alpha emitters in the lungs lead to damage (and later lung cancer) as they are in direct contact with internal organs.
Radon-222 is a radioactive gas that has a high natural abundance (it is a product of the decay of uranium). It is an alpha emitter and the greatest source of naturally occurring radiation exposure.
Beta Radiation
Beta radiation can be either negative (electrons) or positive (positrons). However, in general when people talk about beta radiation or beta particles they are talking about electrons (not positrons). Beta(-) particles are also a form of ionizing radiation (they will ionize molecules in your body) as they are very fast moving electrons. Positrons that are emitted rapidly encounter other electrons and annihilate in reaction that generates gamma radiation. Beta radiation is fairly easily shielded as beta particles have medium penetrating power. They will pass through paper (and skin) but not things like a few millimeters of metal (or most building materials).
Gamma Radiation
Gamma radiation is extremely high energy electromagnetic radiation. Gamma radiation is associated with almost all forms of radioactive decay. As the EM radiation has no associated mass or charge numbers, it is often left out of balanced equations of nuclear change. However, it is a large way by which the energy of a nuclear change is released. Nearly all nuclear decays include emission of gamma radiation.
Gamma radiation has an extremely high penetrating power. Gamma rays will pass through not only paper (or skin) but also most building materials. Effective shielding of gamma rays requires large amounts of thick shielding with heavy metal elements (such as lead). As such, it is very difficult to protect against gamma radiation.
Because gamma radiation is ubiquitously associated with radioactive decay and it the most difficult to shield against, it is the source of most of the damage from radiation. If you have radiation sickness from exposure to radioactive materials, it is almost certainly from gamma rays.
Quantifying Radiation
There are two ways to general ways to quantify amounts of radiation.
1. The amount of "radioactivity" of a given sample.
2. The amount of "radiation" that has been absorbed by a person.
The first of these, the amount of radioactivity of a given sample is measured by counting the number of radioactive decay events that occur for that sample per unit time. This is quantified in one of two units. The old unit the Curie (Ci) which is 3.7 x 1010 disintegrations (decays, events,...) per second. Or in the new unit the Becquerel (Bq) which is 1 event per second. As such, 1 Ci = 3.7 x 1010 Bq. As you might guess a radioactive sample might have a lot of decays in a second. One gram of 226Ra emits has about 1 Ci of activity. So if you use Bq you have lots of big numbers.
The second quantification is not how much radiation a sample has, but how much radiation have you been exposed to. Because many types of radiation are different we have developed scales which try to unify them all into the same measurement. We convert the energy of the different types of radiation into an "effective energy". Then the question is how much of this effective energy have you absorbed. Since the unit typically related to "damage" it is also converted to energy absorbed per kilogram since a large person can absorb more radiation without any effects compared to a small rodent. The units used for this also have an old and new version. The new version is the Sievert (Sv) and corresponds to 1 "effective" J per Kg. The old unit is the rem or "Roentgen Equivalent Man". I00 rem = 1 Sv.
There are two other related expsoure units that quantify the amount of radiation absorbed per kilogram. These reflect simply the total energy absorbed rather than the difference in ionizing power of different types of radiation. They are the rad which is the old unit, and the Gray (Gy) that is the new SI unit.
Quantifying RadiationSources of Radiation
Nuclear radiation is everywhere both from natural and artificial sources. Generally speaking the majority of the radiation people are typically exposed to in their day to day life is due to natural sources. Specifically, Radon gas is the source of the vast majority of the radiation on earth.
A typical exposure for a person over the course of a year is between 200-350 mrem (or 2-3.5 mSv). The specific number depends on the types of activities you under take. If you have lots of x-rays that will increase the number. If you fly in planes a lot that will add some since you'll be exposed to more gamma rays. If you smoke this will greatly increase your radiation exposure since you are breathing smoke into your lungs which is sure to have small amounts of natural radiation.
Other details can be found in the video that follows
Sources of RadiationKinetics and Half Life
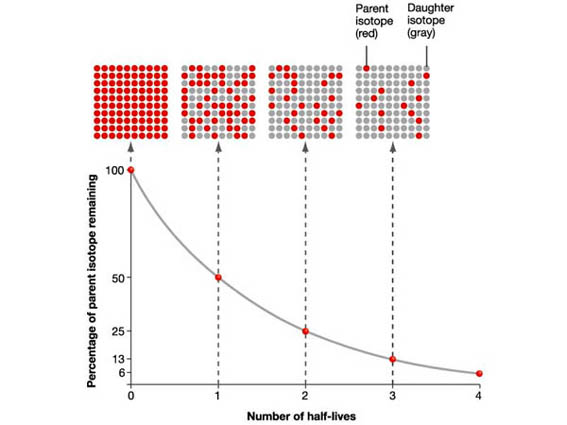
The half-life of a radioactive compound is the time it takes for half of the compound to decay. Because radioactive decay is a first order process (the number of nuclei that decays is proportional to the number of nuclei you have), the half-life is independent of the total number (or concentration).
Thus the half-life is a great description of a radioactive sample as it is independent of the amount of the sample.
The graph depicts the number of radioactive isotope present as a function of time (looking at the population after several half lives). After one half-life, half of the sample has decayed. After another half-life, another half has decayed. So now there is only 25% of the original number of isotopes. After another half-life, half of the remaining isotope decays so you have only 12.5%....
The rate of decay of a nuclear compound follows what is called first order kinetics. The means that the rate of change is proportional the amount of the compound. This proportionality leads to an exponential decay of the amount of radioactive material. Thus over time the rate of radiation emitted from a radioactive compound decays exponentially.
Nuclear Decay Kinetics© 2013 mccord/vandenbout/labrake